 There are lots in my calculations book if you have a copy. That's because there is a decrease in the total number of gas molecules present.Īnd that is all there is to it! You will, of course, need to practise doing this until you are completely confident, but you will need to find your own examples. The entropy has decreased - as we predicted it would in the earlier page. Total entropy at the end = 214 2(69.9) = 353.8 J K -1mol -1Įntropy change = what you end up with - what you started with.Įntropy change = 353.8 - 596 = -242.2 J K -1mol -1 You ended up with 1 mole of carbon dioxide and two moles of liquid water. You started with 1 mole of methane and 2 moles of oxygen. In the introductory page we looked at the following reaction and worked out that there would be a decrease in entropy. Where Σ (sigma) simply means "the sum of". Change in entropy = what you end up with - what you started with You add up the entropies for everything you end up with, and take away the entropies of everything you started with. 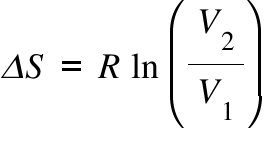
Working out entropy changes for a reaction is very easy. In an exam, you will be given values for all the standard entropies you need. The thing you must be most careful about is the fact that entropy is measured in joules, not kilojoules, unlike most of the other energy terms you will have come across. Use whatever units the examiners give you. 1 bar is 100 kPa 1 atmosphere is 101.325 kPa. Don't worry about it - they are nearly the same. You might find the pressure quoted as 1 atmosphere rather than 1 bar in less recent sources. If your syllabus doesn't mention all these different sorts, just ignore this comment.Įntropy is given the symbol S, and standard entropy (measured at 298 K and a pressure of 1 bar) is given the symbol S°. Entropy change to the surroundings and the total entropy change are dealt with on another page. This page deals only with entropy changes to the system. Note: If you haven't already read the page about introducing entropy, you should do so before you go on. 
We get the cool result that the change in the entropy, $ΔS = S_2 - S_1= k_BN \ln 2$, is proportional to $N$ (so it's extensive) and is independent of the size of $M$, so the change doesn't depend on how big are the little boxes we break the big box into.This page looks at how you can calculate entropy changes during reactions from given values of entropy for each of the substances taking part. We get the new number of microstates and the entropy to be The $N$ molecules can now be put into $2M$ different small volumes. Now pull out the partition so the molecules spread to both parts. We can fix this, but it's not really worth it. But if we have a dilute gas, there is a lot of space, and very few of our boxes will have more than one atom in it. We should really divide by the number of permutations of the atoms in each box. Now this isn't quite right, since if we put more than one atom into a box it doesn't matter in which order we put it in. We can do this for each molecule, so the total number of ways we can put the molecules into the bins is $M \times M \times M. We can put one molecule into the left side of the box into the box in M ways. Let's first calculate the entropy of the initial state. 
(Small compared to the size of the box, but large compared to the size of an atom, so we don't have to worry about atoms "filling up one of the small volume".) One way to do this is to imagine breaking up the left side of the box into M small volumes. When the partition separating the two halves of the box is removed and the system reaches equilibrium again, how does the new entropy of the gas compare to the energy of the original system? An ideal gas occupies half of the container and the other half is empty. Presenting a sample problemĬonsider an example of an isolated box of volume $2V$ divided into two equal compartments. 
We choose a model that is simple enough that we can see all the details. Entropy change calculator free#(This example will be of particular interest when we start thinking about free energy.) Now, let's look at a toy model example that lets us see how the microstate counting form of entropy, $S = k_B \ln W$, can also give us insights into what happens spontaneously. In our analysis of the entropy change in heat flow, we analyzed a toy model to see how the macroscopic form of entropy, $S = Q/T$, gave us insight into what would happen spontaneously. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
